The greenhouse effect is a naturally occurring process that aids in heating the Earth's surface and atmosphere. It results from the fact that certain atmospheric gases, such as carbon dioxide, water vapor, and methane, are able to change the energy balance of the planet by absorbing longwave radiation emitted from the Earth's surface. Without the greenhouse effect life on this planet would probably not exist as the average temperature of the Earth would be a chilly -18° Celsius, rather than the present 15° Celsius.
As energy from the Sun
passes through the atmosphere a number of things take
place (see Figure
7h-1). A portion of the energy (26% globally)
is reflected or scattered back
to space by clouds and other atmospheric particles. About
19% of the energy available is absorbed by clouds, gases
(like ozone),
and particles in the atmosphere. Of the remaining 55%
of the solar energy passing through the Earth's atmosphere,
4% is reflected from the surface back to space. On average,
about 51% of the Sun's radiation reaches the surface.
This energy is then used in a number of processes, including
the heating of the ground surface; the melting of ice
and snow and the evaporation of water; and plant photosynthesis.
The heating of the ground by sunlight causes the Earth's surface to become
a radiator of energy in the longwave band (sometimes called infrared
radiation). This emission of energy is generally directed to space
(see Figure 7h-2). However, only a small portion
of this energy actually makes it back to space. The majority of the outgoing
infrared radiation is absorbed by the greenhouse
gases (see Figure 7h-3 below).
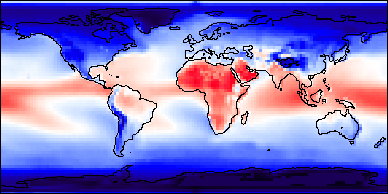

Figure 7h-3: Annual (1987) quantity of outgoing longwave radiation absorbed in the atmosphere.
(Image created by the CoVis Greenhouse Effect Visualizer).
Absorption of longwave radiation by the atmosphere causes additional heat energy to be added to the Earth's atmospheric system. The now warmer atmospheric greenhouse gas molecules begin radiating longwave energy in all directions. Over 90% of this emission of longwave energy is directed back to the Earth's surface where it once again is absorbed by the surface. The heating of the ground by the longwave radiation causes the ground surface to once again radiate, repeating the cycle described above, again and again, until no more longwave is available for absorption.
The amount of heat energy added to the atmosphere by the greenhouse effect is controlled by the concentration of greenhouse gases in the Earth's atmosphere. All of the major greenhouse gases have increased in concentration since the beginning of the Industrial Revolution (about 1700 AD). As a result of these higher concentrations, scientists predict that the greenhouse effect will be enhanced and the Earth's climate will become warmer. Predicting the amount of warming is accomplished by computer modeling. Computer models suggest that a doubling of the concentration of the main greenhouse gas, carbon dioxide, may raise the average global temperature between 1 and 3° Celsius. However, the numeric equations of computer models do not accurately simulate the effects of a number of possible negative feedbacks. For example, many of the models cannot properly simulate the negative effects that increased cloud cover would have on the radiation balance of a warmer Earth. Increasing the Earth's temperature would cause the oceans to evaporate greater amounts of water, causing the atmosphere to become cloudier. These extra clouds would then reflect a greater proportion of the Sun's energy back to space reducing the amount of solar radiation absorbed by the atmosphere and the Earth's surface. With less solar energy being absorbed at the surface, the effects of an enhanced greenhouse effect may be counteracted.
A number of gases are involved
in the human caused enhancement of the greenhouse effect
(see Table
7h-1 below). These gases include: carbon
dioxide (CO2); methane (CH4); nitrous
oxide (N2O); chlorofluorocarbons (CFxClx);
and tropospheric
ozone (O3). Of these gases, the single
most important gas is carbon
dioxide which accounts for about 55% of the
change in the intensity of the Earth's greenhouse effect.
The contributions of the other gases are 25% for chlorofluorocarbons,
15% for methane,
and 5% for nitrous
oxide. Ozone's contribution
to the enhancement of
greenhouse effect is still yet to be quantified.
Average concentrations of atmospheric carbon
dioxide in the year 2005 were about 380 parts per million (see Figure
7a-1). Prior to 1700, levels of carbon dioxide were about 280
parts per million. This increase in carbon dioxide in the atmosphere is primarily
due to the activities of humans. Beginning in 1700, societal changes brought
about by the Industrial
Revolution increased
the amount of carbon dioxide entering the atmosphere. The major sources of
this gas include fossil fuel combustion for industry, transportation, space
heating, electricity generation and cooking; and vegetation changes in natural
prairie, woodland, and forested ecosystems. Emissions from fossil fuel combustion
account for about 65% of the extra carbon dioxide now found in our atmosphere.
The remaining 35% is derived from deforestation and the conversion of prairie,
woodland, and forested ecosystems primarily into agricultural systems. Natural
ecosystems can hold 20 to 100 times more carbon dioxide per unit area than
agricultural systems.
Artificially created chlorofluorocarbons are
the strongest greenhouse gas per molecule. However, low concentrations in the
atmosphere reduce their overall importance in the enhancement of
the greenhouse effect. Current measurements in the atmosphere indicate that
the concentration of these chemicals may soon begin declining because of reduced
emissions. Reports of the development of ozone holes over the North and South
Poles and a general decline in global stratospheric ozone levels over the last
two decades has caused many nations to cutback on their production and use
of these chemicals. In 1987, the signing of the Montreal
Protocol agreement by forty-six nations established an immediate timetable
for the global reduction of chlorofluorocarbons production and use.
Since 1750, methane concentrations
in the atmosphere have increased by more than 150%. The primary sources for
the additional methane added to the atmosphere (in order of importance) are
rice cultivation, domestic grazing animals, termites, landfills, coal mining,
and oil and gas extraction. Anaerobic conditions associated with rice paddy
flooding results in the formation of methane gas. However, an accurate estimate
of how much methane is being produced from rice paddies has been difficult
to obtain. More than 60% of all rice paddies are found in India and China
where scientific data concerning emission rates are unavailable. Nevertheless,
scientists believe that the contribution of rice paddies is large because this
form of crop production has more than doubled since 1950. Grazing animals release
methane to the environment as a result of herbaceous digestion. Some researchers
believe the addition of methane from this source has more than quadrupled over
the last century. Termites also release methane through similar processes.
Land-use change in the tropics, due to deforestation, ranching, and farming,
may be causing termite numbers to expand. If this assumption is correct, the
contribution from these insects may be important. Methane is also released
from landfills, coal mines, and gas and oil drilling. Landfills produce methane
as organic wastes decompose over time. Coal, oil, and natural gas deposits
release methane to the atmosphere when these deposits are excavated or drilled.
The average concentration of nitrous
oxide in the atmosphere is now increasing at a rate of 0.2 to 0.3%
per year. Sources for this increase include land-use conversion; fossil fuel
combustion; biomass burning; and soil fertilization. Most of the nitrous oxide
added to the atmosphere each year comes from deforestation and the conversion
of forest, savanna and grassland ecosystems into agricultural fields and rangeland.
Both of these processes reduce the amount of nitrogen stored in living vegetation
and soil through the decomposition of organic matter. Nitrous oxide is also
released into the atmosphere when fossil fuels and biomass are burned. However,
the combined contribution of these sources to the increase of this gas in the
atmosphere is thought to be minor. The use of nitrate and ammonium fertilizers
to enhance plant growth is another source of nitrous oxide. Accurate measurements
of how much nitrous oxide is being released from fertilization have been difficult
to obtain. Estimates suggest that the contribution from this source may represent
from 50% to 0.2% of nitrous oxide added to the atmosphere annually.
Ozone's role
in the enhancement of the greenhouse effect has been difficult
to determine scientifically. Accurate measurements of past long-term
(more than 25 years in the past) levels of this gas in the atmosphere
are currently unavailable. Concentrations of ozone gas are found
in two different regions of the Earth's atmosphere. The majority
of the ozone (about 97%) found in the atmosphere is localized
in the stratosphere at
an altitude of 15 to 55 kilometers above the Earth's surface. In
recent years, the concentration of the stratospheric
ozone has been decreasing because of the buildup of chlorofluorocarbons in
the atmosphere (see Lecture 7e). Since
the late 1970s, scientists have discovered that total
column ozone amounts over Antarctica in the springtime
have decreased by as much as 70%. Satellite measurements have
indicated that the zone from 65° North to 65° South latitude
has had a 3% decrease in stratospheric ozone since 1978. Ozone
is also highly concentrated at the Earth's surface. Most of this
ozone is created as an artificial by product of photochemical
smog.
In summary, the greenhouse effect causes the atmosphere to trap more heat energy at the Earth's surface and within the atmosphere by absorbing and re-emitting longwave energy. Of the longwave energy emitted back to space, 90% is intercepted and absorbed by greenhouse gases. Without the greenhouse effect the Earth's average global temperature would be -18° Celsius, rather than the present 15° Celsius. In the last few centuries, the activities of humans have directly or indirectly caused the concentration of the major greenhouse gases to increase. Scientists predict that this increase may enhance the greenhouse effect making the planet warmer. Some experts estimate that the Earth's average global temperature has already increased by 0.3 to 0.6° Celsius, since the beginning of this century, because of this enhancement. Predictions of future climates indicate that by the middle of the next century the Earth's global temperature may be 1 to 3° Celsius higher than today.
Table 7h-1: Gases involved in the Greenhouse Effect: past and present concentration and sources.
|
Greenhouse Gas |
Concentration 1750 |
Concentration 2003 |
Percent Change |
Natural and Anthropogenic Sources |
|---|---|---|---|---|
| Carbon Dioxide | 280 ppm | 376 ppm | 34% | Organic decay; Forest fires; Volcanoes; Burning fossil fuels; Deforestation; Land-use change |
| Methane | 0.71 ppm | 1.79 ppm | 152% | Wetlands; Organic decay; Termites; Natural gas & oil extraction; Biomass burning; Rice cultivation; Cattle; Refuse landfills |
| Nitrous Oxide | 270 ppb | 319 ppb | 18% | Forests; Grasslands; Oceans; Soils; Soil cultivation; Fertilizers; Biomass burning; Burning of fossil fuels |
| Chlorofluorocarbons (CFCs) | 0 | 880 ppt | Not Applicable | Refrigerators; Aerosol spray propellants; Cleaning solvents |
| Ozone | Unknown | Varies with latitude and altitude in the atmosphere | Global levels have generally decreased in the stratosphere and increased near the Earth's surface | Created naturally by the action of sunlight on molecular oxygen and artificially through photochemical smog production |